Sign up for the daily CJR newsletter.
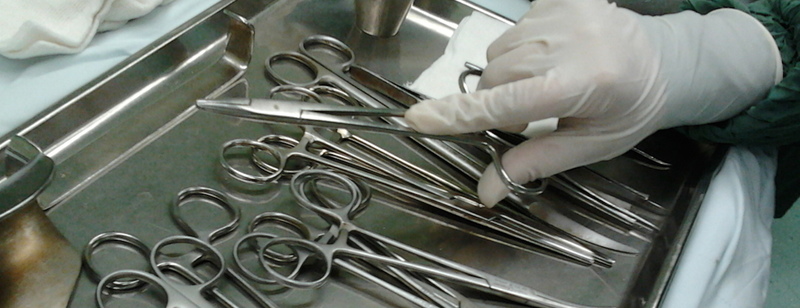
Stories about medical dangers often rely on anecdotes about specific patients at risk. But in a recent investigative series, reporters Karen Bouffard and Joel Kurth of The Detroit News showed another way to tell a compelling, high-impact narrative: Backed by key documents and interviews, they demonstrated persistent, systemic problems with dirty surgical instruments at a hospital network in the city. The series has prompted a joint state-federal investigation, spurred a flurry of coverage in other Detroit news outlets, and could galvanize an important discussion about public disclosure of hospital safety data.
In the first article of the series, published August 26, Bouffard and Kurth get right to the point: They have about 200 pages of emails and reports that depict years of complaints about dirty instruments from staffers and surgeons at Detroit Medical Center’s Midtown campus:
The records show improperly sterilized tools complicated operations from appendectomies and brain surgeries to cleft palate repair and spinal fusions. Patients were kept under anesthesia for up to an hour as staffers replaced instruments. Dozens of operations were canceled at the last minute, some after anesthesia was administered.
At least twice a child’s chest or skull was open for surgery when doctors discovered dirty instruments. In January 2015, open-heart surgery for a 7-month-old girl was interrupted at Children’s Hospital of Michigan because a tube leading to a bypass machine was clogged with blood from a previous operation.
Those details grab your attention, but what the reporting drives home is how long the concerns persisted, even as doctors and other staffers repeatedly sounded the alarm. The News has Laura Cortner, a former director of the center’s Central Sterilization Department, saying that “huge challenges” have lasted for years with “no prolonged effort” to fix them. In a memo from mid-2015, highlighted in a timeline accompanying the story, the chief surgeon at Children’s Hospital tells top hospital officials, “We are putting patients at risk frequently and now canceling up to 10 cases this week…promises just aren’t cutting it.” The News even has the center’s chief administrator on record, saying, “This is something that has to be fixed.” (The center has also said that “no safety issues or surgical site infections” actually resulted from unsterile instruments, and has pointed to its efforts to address the concerns. A spokesperson for the Detroit Medical Center said in a statement to CJR that the center “took an important step earlier this year when we brought in an industry leading sterile processing management company, and we continue to work closely with them.”)
The News’ investigation, which lasted about six months, took off when the reporters got hold of the email and other records. “It was obvious when we looked at the documents there were system problems. There were so many emails for a long time, and there never seemed to be a resolution,” Bouffard told me in an interview.
“What distinguishes this story is the strength of the emails and the strength of the records showing these systemic failures for 11 years,” Kurth added. “They spoke better to the situation than one anecdote could.”
That first piece did feature one good anecdote—the story of Kalaya Hull-Mason, who underwent heart surgery at Children’s Hospital when she was seven months old. The operation was stopped when a suction tube needed to draw blood appeared to be clogged with blood from a previous patient. The operation was eventually successful after staff covered the baby’s heart, dismantled the bypass machine, and rebuilt the surgical field. One document gave the reporters a clue about Kalaya, and Kurth spent two months off and on tracking down her parents. Though hospital records indicated one of the girl’s parents had been informed of the complications, both told the News they had been unaware of the problems until the paper contacted them.
That gets to one of the other themes the series drives home: the patchwork standards for disclosure to patients and the public at large about risks from medical procedures.
As the News reported, the center declined to provide detailed infection data that would shed light on its overall performance, and it isn’t required to. Learning about the regulatory framework for hospitals, and what information is and isn’t available to the public, was the most time-consuming part of the reporting process, says Bouffard. The second main story in the series offers a good description of how oversight in the medical industry, which is largely self-policing, often isn’t transparent to the wider world. State and federal agencies collect a substantial amount of data, but let hospitals supply it voluntarily on the condition that much of it will be kept confidential. There’s also no legal requirement in Michigan that doctors have to inform patients of complications, and guidelines about when to do so are subjective. Kurth says, “Doctors don’t have to tell patients. Patients don’t know, so nobody tells the regulators.”
There’s a broader discussion happening in healthcare right now about greater transparency on a number of fronts, and the News series is an important contribution to that debate—in addition to providing important scrutiny that benefits the people of Detroit. Score this on a hit for accountability coverage by a local newspaper.
Has America ever needed a media defender more than now? Help us by joining CJR today.